Sex, immunity, and the brain

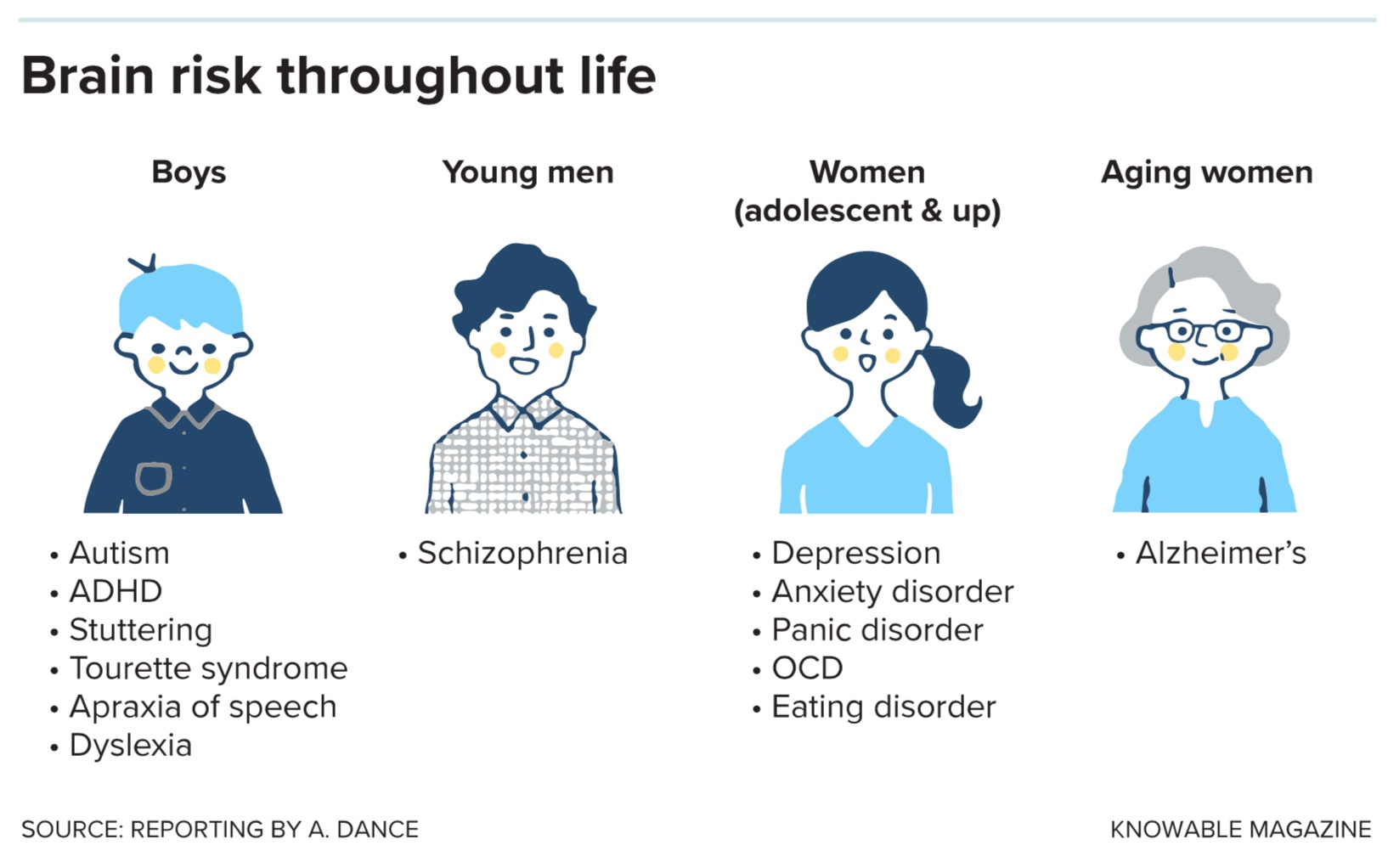
Suppose a couple has two children, a boy and a girl. Chances are, they’ll both grow up with typical, healthy brains. But should either diverge from the usual route of brain development, or suffer mental health issues, their paths are likely to be different.
The son’s differences might show up first. All else being equal, he’s four times more likely than his sister to be diagnosed with autism. Rates of other neurodevelopmental conditions and disabilities are also higher in boys. As he grows into a young man, his chances of developing schizophrenia will be two to three times higher than hers.
When the siblings hit puberty, those relative risks will flip. The sister will be almost twice as likely to experience depression or an anxiety disorder. Much later in life, she’ll be at higher risk of developing Alzheimer’s disease.
Those trends are not hard and fast rules, of course: Men can and do suffer from depression and Alzheimer’s; some girls develop autism; women aren’t immune to schizophrenia. Male and female brains are more alike than they are different.
But scientists are learning that there’s more to these different risk profiles than, say, the pressures women face in a patriarchal society or the fact that women tend to live longer, giving diseases of aging time to develop. Subtle biological differences between male and female brains, and bodies, are important contributors.
To explain these sex differences, there are some obvious places to look. The female’s two X chromosomes, to the male’s single copy, is one. Differing sex hormones — primarily testosterone in males and estrogen in females — is another. But a steadily growing body of research points to a less obvious influence: the cells and molecules of the immune system.
Scientists have long had of evidence linking immune activity to brain differences and disorders, but the science that sex adds to that equation is still developing. Until the past decade, neuroscientists routinely used only male animals in their experiments, fearing that female hormonal cycles would interfere with results. This turned out to be much less of an issue than originally thought. Moreover, scientists now know that hormones in male rodents can fluctuate just as much — not on a set cycle, but in response to factors like their position in the social hierarchy of their cage group. Since 2016, the National Institutes of Health has asked applicants for research funds to use both sexes or explain why they’re only using one.
In recent studies, neuroscientists have found that immune cells called microglia work differently in the developing brains of male and female rodents, even in the absence of any infection. These microglial actions, reflected in studies of people, may predispose boys to neural differences and disorders early in life, the researchers speculate, but could protect them later on. Scientists have also identified several genes involved in immune responses that could help to explain heightened risks for girls and women from puberty onward. With time, a better understanding of these differences might lead to sex-specific treatments.
“We’re just starting to carve into this,” says Justin Bollinger, a neuroscientist at the University of Cincinnati. “It’s super important and super sad that for the longest time, researchers felt that males were sufficient, that males and females acted the same, that they responded to the same things.”
Immunity in the developing brain
One of the first clues linking brain development and immune responses came in the late 1980s, when researchers examined birth records and psychiatric hospital records in Finland, where there had been an influenza epidemic in the fall of 1957. The scientists found that if pregnant women had been in their second trimester that autumn, their adult children were about 50 percent more likely to be admitted to the hospital with a schizophrenia diagnosis than were children of women who had been in their first or third trimester during the epidemic.
Other studies supported this finding, suggesting that if a woman’s immune system is called upon to fight off an infection during pregnancy, it can predispose her offspring to schizophrenia. “That’s really brought a lot of attention to how the immune system can make the developing brain go awry,” says Margaret McCarthy, a neuroscientist at the University of Maryland School of Medicine in Baltimore. Researchers in New York, meanwhile, documented a variety of neurological challenges in the children of mothers who contracted rubella during a 1964 outbreak, including an unusually high rate of autism.
To mimic the effects of outbreaks in human populations and investigate possible mechanisms, scientists have injected pregnant rats and mice with noninfectious pieces of bacteria or viruses. This causes an immune response in the mother, which in turn influences immune activity in the offspring. Then the researchers study the pups after they’re born.
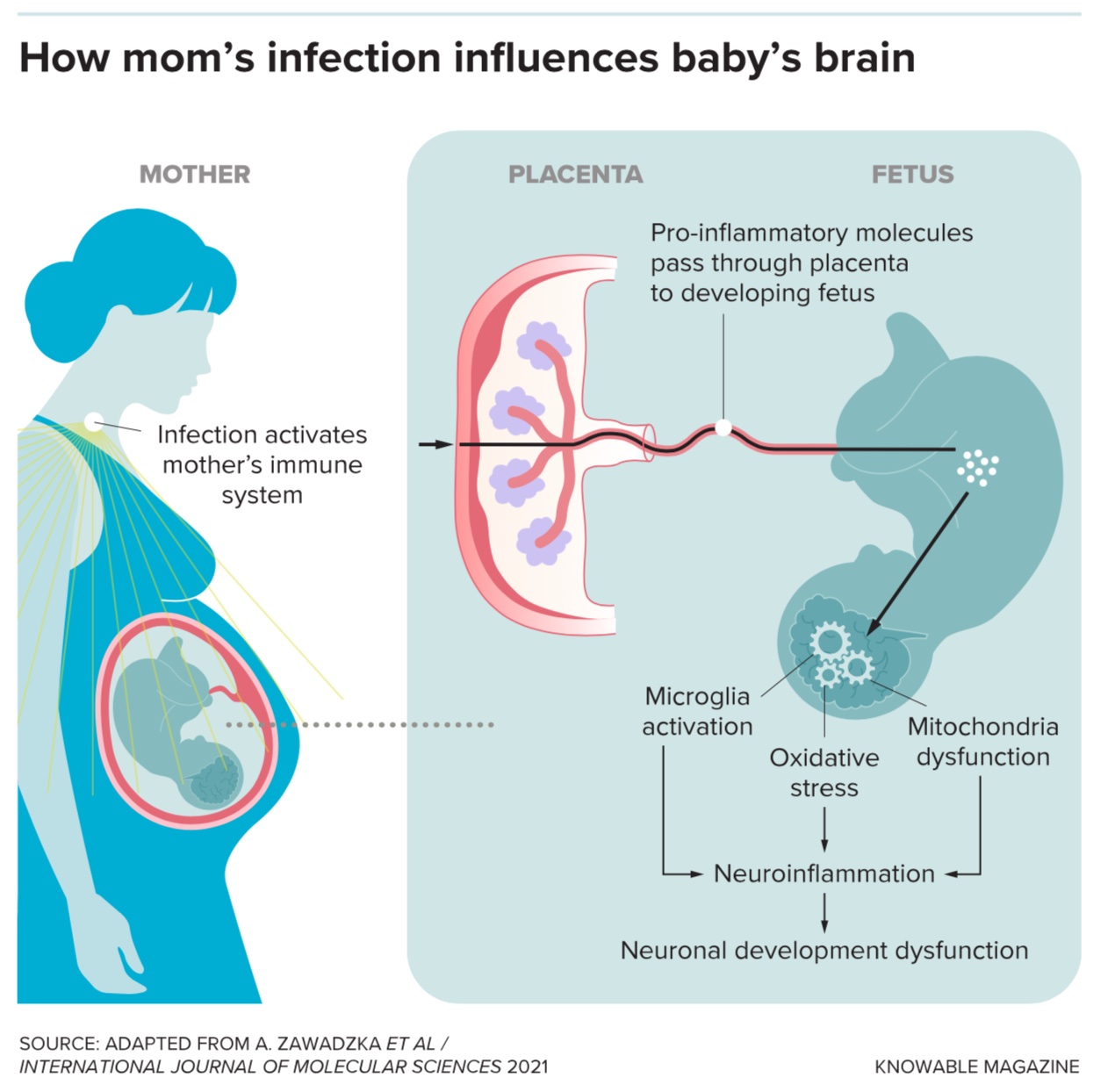
These studies have backed up the idea that maternal infection affects the baby’s brain. While it’s difficult to say if a rodent is experiencing specific signs of autism or schizophrenia, scientists do observe that the pups are more anxious and less social than those birthed by mothers who didn’t experience an immune challenge.
The pups also have more — and more active — microglia. These cells, which make up 10 percent of the brain, are the organ’s resident immune cells: Their job is to swallow invading bacteria, viruses and fungi, as well as to consume regular cellular garbage. But they do much more than that. Microglia also release chemicals known as growth factors to support the brain. And during fetal development, they break down unnecessary connections between nerve cells, or even eliminate cells entirely — actions that sculpt the brain’s wiring. Should microglia be exposed to infection during this critical time, some scientists suggest that sculpting might go awry and the brain might bear long-term consequences.
So far, the evidence for these effects is more limited in humans, but brain scans and autopsy studies both find unusually high numbers of active microglia in people with schizophrenia or autism.
Male and female microglia
Sex adds another wrinkle to the link between microglia and brain development: These cells behave differently in certain parts of developing male and female brains even when all is going normally.
Take, for example, McCarthy’s 2019 findings on young rats at play. Play in young animals is influenced by a brain region called the amygdala, and it’s known that the effect of testosterone on the amygdala biases male pups toward rough-and-tumble play — a tendency not seen in females. The researchers found that microglia in the amygdala were more active in the males, due to that testosterone exposure in the uterus. The highly active male microglia ate up another type of cell that develops into star-shaped cells called astrocytes. “They’re basically engaging in cellular murder,” says McCarthy.
In females, the scientists observed, these astrocytes survive and seem to dampen the activation of amygdala nerve cells — and this dampening, in turn, seems to reduce roughhousing. And when the researchers used an antibody to prevent microglia from killing the relevant astrocytes in male pups, the pups no longer played rough. McCarthy says the astrocytes, when present, suppress the activity of nerves in real time to prevent rough play.
There are many other reported ways that microglia act differently in typical male and female rodent brains at a very young age. And those differences can have long-term consequences, particularly if the mother encounters an infection or if scientists mimic one in the lab. For example, the authors of a 2020 study looked at microglia in the adult offspring of mouse mothers exposed to a synthetic molecule that mimics the genetic material of a virus. They focused on a part of the brain called the dentate gyrus, a region involved in learning and memory that is typically smaller in people with schizophrenia.
In the study, male mice born to mothers treated with the viral mimic had a greater density of synapses — connections between nerve cells — than typical in the dentate gyrus. This was true both for excitatory synapses, where one neuron excites activity in the next, and for inhibitory synapses, where one neuron dampens the activity of another. In females, in contrast, the viral mimic treatment resulted in fewer excitatory synapses, and little change to the inhibitory ones. These changes to the proportions of “on” and “off” synapses bear similarities to synapse imbalances seen in human schizophrenia and in addition suggest that the pattern differs between males and females.
Based on this and other research, the working hypothesis is that immune activation in the brain, very early in life, changes microglia and somehow “primes” the brain for differences that emerge later on. Exactly how this might work isn’t clear, says Jaclyn Schwarz, a neuroscientist at the University of Delaware in Newark. One possibility is that microglia in males, distracted by an infection, skip some of the pruning work they normally do during fetal development. Alternatively, she speculates, perhaps the male microglia become hyperactive over the long term, pruning away too many neural connections as the brain continues to develop during childhood and into adolescence and adulthood.
The female brain
In human teenagers, the overall pattern of sex differences in mental illness reverses. Women and adolescent girls are more susceptible to mood disorders like depression and anxiety, which have less to do with the brain’s wiring during development and more to do with ongoing chemical processes within the brain, says Schwarz.
The brain isn’t the only place the immune system differs by sex: Women frequently mount a stronger immune response to infections than males do. When neuroscientists were studying only male rodents, immunologists, in contrast, often focused on female animals and their cells because they offer a more robust response, says Natalie Tronson, a behavioral neuroscientist at the University of Michigan in Ann Arbor. Women pay a price for that powerful response with a higher rate of autoimmune conditions like lupus.
Researchers have analyzed the genes turned on and off in brain tissue of people who had depression, and they’ve found that the patterns of gene use differ by sex. “What’s happening in the brain of a man or woman with depression is very different,” says Georgia Hodes, a neuroscientist at Virginia Tech in Blacksburg and a coauthor of that research. One pattern they’ve seen in women is changes to the activity of genes involved in inflammation, a key immune mechanism.
Brain inflammation is also closely tied to Alzheimer’s disease, and sex influences risk there too: Early symptoms typically arise in one’s 60s and women are more likely than men to be diagnosed. In a 2021 study, Marina Sirota, a bioinformaticist at the University of California, San Francisco, and colleagues examined which genes were turned on or off in brains of people who died with Alzheimer’s. They discovered alterations in gene activity that would influence immune activity in women who had Alzheimer’s compared to women who didn’t. They saw no such difference in men.
Sirota says there’s more work to do to understand why these gene patterns change in women, and how they might influence the course of dementia. (As it happens, integrating such complex immunity data is her specialty; she recently penned a review on the topic for the Annual Review of Biomedical Data Science.) As for older men, the microglia that can cause increased risk during development might turn out to be beneficial in old age. Another study of brain tissue from people who had Alzheimer’s found that the microglia of men’s brains were more likely to adopt an amoeba-like shape, associated with protective activity, than were microglia in women.
Mouse studies indicate that there are differences even between healthy male and female brains as animals age. Scientists led by Bill Freeman, a neuroscientist at the Oklahoma Medical Research Foundation in Oklahoma City, examined the patterns of genetic activity and proteins in the brains of two-year-old mice (that’s a ripe old age for a mouse). They saw that while inflammation increased with age in the brains of both sexes, the change was more prominent in females.
Thus, while studies of males point to microglia as major players in both early risk and later protection, the situation in women seems to involve a host of immune genes that may influence risk for inflammation, mood disorders and dementia in a yet-to-be-understood manner.
A step toward health equity
Scientists can only speculate on why these differences evolved, but many point to the simple fact that females get pregnant. The mother’s immune system must not attack the fetus, even though it differs genetically from her own body. Thus, pregnancy causes a number of changes to immunity, some of which muffle a mother’s immune system, making her more prone to severe illness from some infections, such as Covid-19 and chickenpox.
“Men don’t seem to have that flexibility to their immune system,” says Hodes. “They always are having the same immune responses throughout their lifespan, with some changes in aging.” But the female immune flexibility — needed to protect the fetus — may come at a cost to the female brain.
No one’s designing pink and blue pills just yet. But cataloging such differences is an important first step in understanding how sex, the brain and the immune system interact in health and disease. Getting to the root of these differences, and ultimately developing different treatments for different people, is crucial to achieving health equity.
“We are trying to understand biology, we’re trying to improve health,” says Freeman. “That means understanding the diversity of our human species.”
This article originally appeared in Knowable Magazine, a nonprofit publication dedicated to making scientific knowledge accessible to all. Sign up for Knowable Magazine’s newsletter.