Most of the seafloor is a thick graveyard of ooze

- The majority of the bottom of the ocean is covered in morbid ooze, hundreds of feet deep.
- This substance is made of the skeletons of an uncountable number of tiny creatures, raining down from above.
- The passage of material through this ooze is a substantial part of several biological, chemical, and geological cycles.
We often picture the bottom of the sea as a graveyard for ships. They lie in rest on a bed of rocks and sand, where angelfish nibble at shiny doubloons and sharks nibble at greedy divers. More common on the ocean floor, however, is a graveyard of another sort: The bodies of an unfathomable number of microscopic creatures form an ooze, hundreds of feet deep.
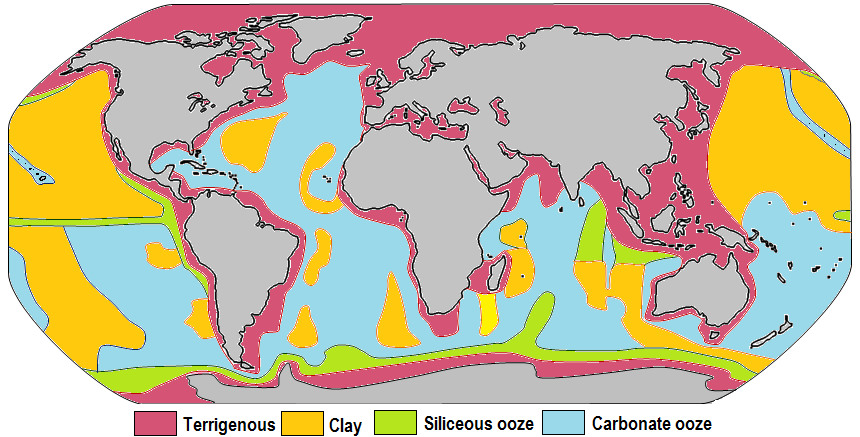
Oozes are a type of marine sediment, differentiated from clay or mud by being made 30% or more from shells, skeletons, and pieces of the dead. There are two main sorts of ooze, each with different embodiments. Combined, they cover the majority of Earth’s seafloor.
Siliceous oozes
Each year, several trillion pounds of microscopic silicon-based skeletons fall down the water column to pile up into siliceous ooze. This is part of a global process in which silicates — simple compounds of Si and O — are cycled through the sea annually. New silicates are introduced through geological erosion and brought into the biological realm by plankton, a catch-all term for an enormous variety of small life forms haplessly drifting about the ocean. Many of these creatures build themselves silicate shells.
Siliceous ooze dominates two broad belts of the seafloor: one surrounding the equator and another at the high latitudes near Antarctica. While the sheer mass of material raining down is large, the layer grows at less than an inch per millennium. That doesn’t sound like much, but imagine the volume of dust required to form an inch-thick layer on your bookshelf. Now imagine the layer covers all of North America and South America.
There are two predominant forms of siliceous ooze.
Radiolarian ooze
Radiolarians are microscopic organisms that fall under the umbrella of plankton. Their body consists of one cell with organelles, and in some cases symbiotic algae, sheltered inside a silicate cage. Roughly the width of two human hairs, the silicate cages are of exquisite geometric composition. The 19th-century zoologist Ernst Haeckel drew a book of beautiful charts of the many radiolarians he discovered under a microscope.
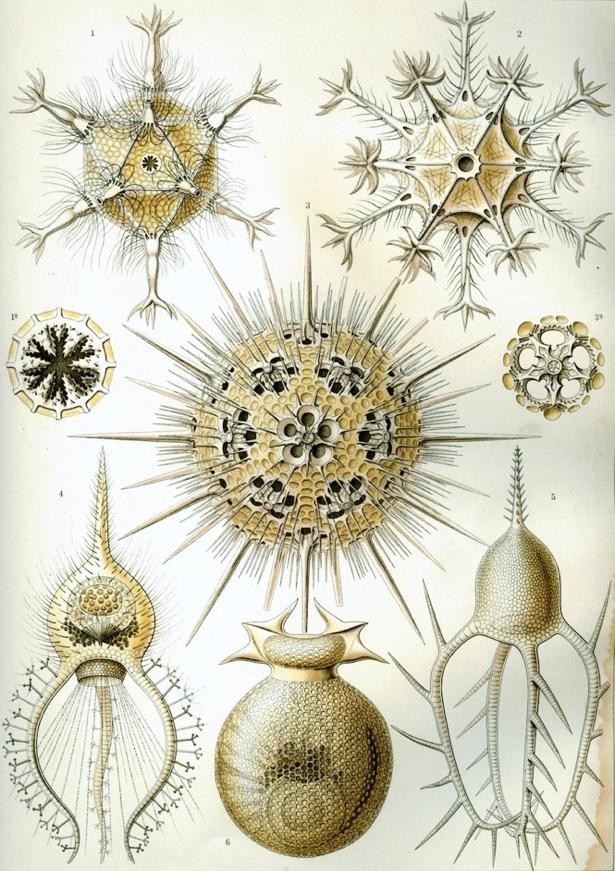
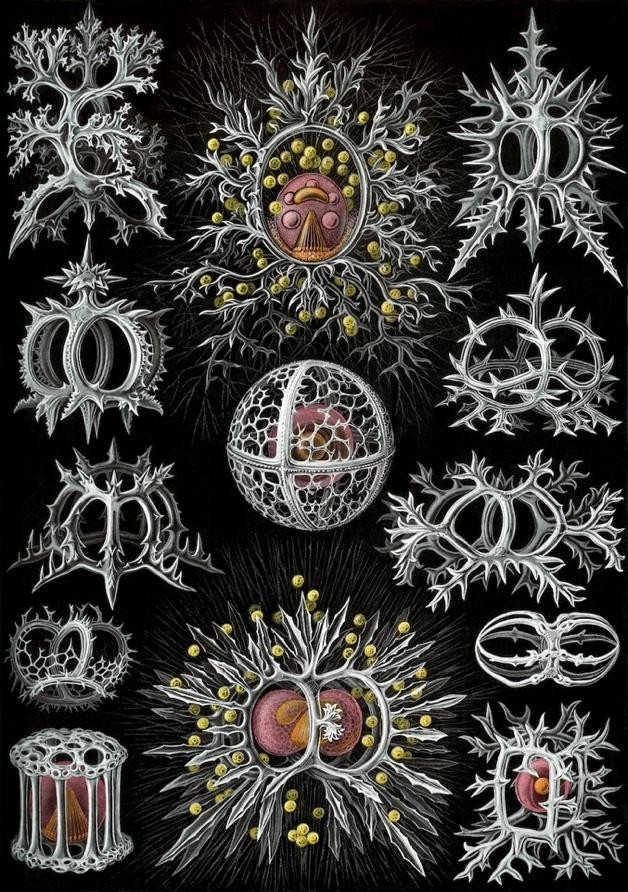
After spending a few weeks photosynthesizing in shallow water, the organisms gradually sink over a period of months. They may come to rest on the seafloor, if it lies above the depth at which their silicate structures dissolve. Radiolarian ooze fossilizes and solidifies into radiolarite, a hard sedimentary rock layer. Paleolithic humans made edged tools from it, while modern humans find other uses.
Diatom ooze
Diatoms are single-celled algae. They, too, form elaborate geometric shells that gleam in microscope images. With lifespans measured in days and reproductive cycles measured in hours, they are, unsurprisingly, staggeringly common in the ocean.
Diatom photosynthesis is believed to produce a substantial fraction of all the oxygen in our atmosphere. Diatom ooze of the distant past, fossilized and tectonically transported into continental landmasses, is an important component of toothpaste, cat litter, and dynamite.

Calcareous oozes
The second major type of ooze is calcareous, meaning composed primarily of calcium carbonate. It is the accumulation of organisms whose shells and exoskeletons are formed of this substance: plankton like microscopic foraminifera, molluscs, and sea sponges.
Over time the shells accumulate, break down, and fuse together to form rocks with which surface-dwellers are familiar, such as limestone and chalk. The passage of calcium carbonate from living creatures, through ooze, into rock is part of a global cycle of carbon, far too complex to describe here.
Calcareous ooze primarily forms in shallower water. In deeper water it is chemically favorable for the carbonate material to dissolve, and the siliceous oozes dominate down to their own dissolution depth. The number of creatures with carbonate shells is so great that despite dissolution claiming a great majority, the sediment layer may grow as quickly as one inch per millennium. Calcareous ooze covers roughly half of the seafloor, a total area comparable to the entire landmass of earth. The total mass of ooze may be on the order of 10 million billion tons.
Globigerina ooze
The single most common seafloor ooze is made of the shells, or tests, of globigerina, coccolithophores, and similar single-celled organisms. Globigerina come in a variety of forms, generally a series of connected spheres and chambers. At less than a millimeter in diameter they’re comparable in size to a single grain of sand.
The living creature is a spiny photosynthetic protist of the biological classification foraminifera, specifically a type that lives floating in the water rather than on the seafloor. Coccolithophores surround their central cell with an exoskeleton composed of a series of circular armor plates called coccoliths. When they die, their calcite shells and coccoliths pile up on the seafloor.
Pteropod ooze
A small proportion of calcareous ooze is formed from the calcium-rich remains of sea slugs, sea snails, nudibranchs, and possibly even stranger things like sea hares. The calcium carbonate polymorph aragonite in their shells is more easily soluble than the calcite polymorph shells forming the globigerina ooze, so pteropod ooze is only common in very shallow water.
We’ve barely scraped the top layer of the subject of pelagic oozes. This biological sedimentation is one part of the complicated cycling of silicate and carbonaceous minerals through the biological and geological strata of the planet. It’s also a monumental tomb for the vast numbers of tiny life forms that shape the living conditions on Earth.